The Periodic Table Is Made Of _____ Known Elements.
sampleletters
Mar 14, 2026 · 7 min read
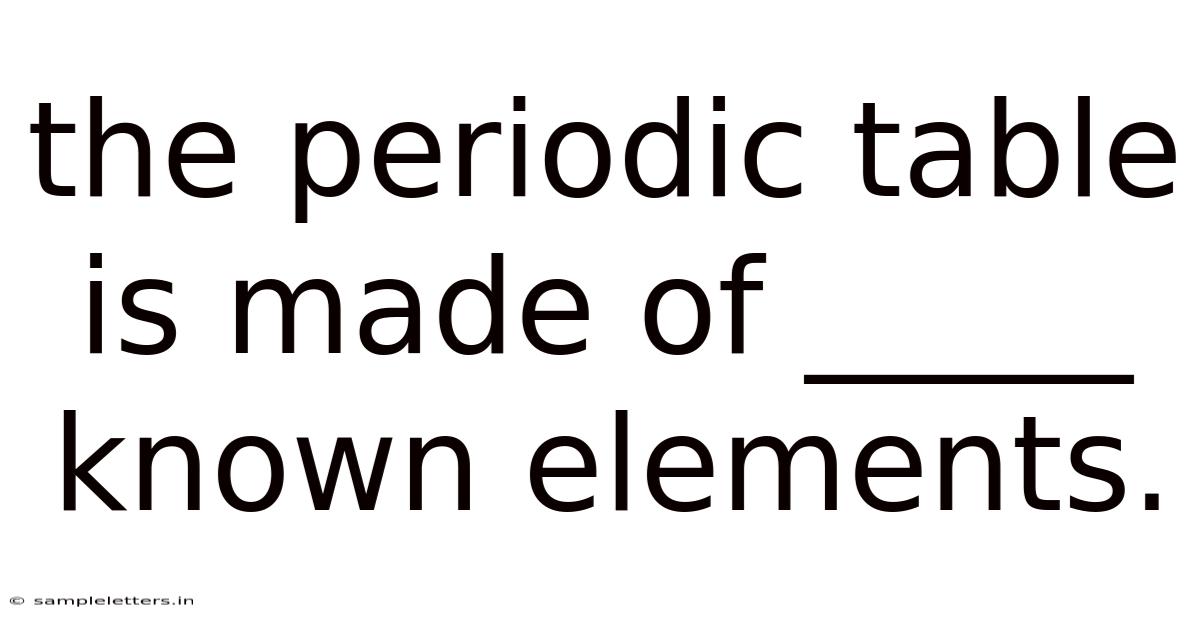
Table of Contents
The Periodic Table Is Made of 118 Known Elements
The periodic table is a systematic arrangement of chemical elements that reveals the underlying order of matter. When we ask, “the periodic table is made of _____ known elements,” the answer is 118. This figure represents the total count of elements that scientists have discovered, confirmed, and officially recognized by the International Union of Pure and Applied Chemistry (IUPAC) as of 2025. Understanding how this number was reached, what it signifies, and how the elements are organized provides a gateway to the fascinating world of chemistry and its many applications.
How the Number 118 Was Determined
- Discovery and Validation – Elements are first synthesized in laboratories or isolated from natural sources. Each new candidate undergoes rigorous verification of its atomic number, decay chain, and physical properties.
- IUPAC Naming Process – Once a collaboration confirms the existence of a new element, they submit a proposal to IUPAC. The governing body reviews the data, and if approved, assigns a permanent name and symbol.
- Periodic Table Update – Every time IUPAC adds a validated element, the periodic table is updated to reflect the new entry. The most recent addition, oganesson (Og, element 118), completed the seventh period in 2016, bringing the total to 118.
These steps ensure that the count of known elements is not arbitrary but grounded in experimental evidence and international consensus.
The Structure Behind the Count
The periodic table is organized into rows (periods) and columns (groups) based on recurring chemical properties. The arrangement reflects electron configurations, which dictate how atoms bond and react.
- Periods run horizontally and indicate the number of electron shells an atom possesses.
- Groups run vertically and share similar valence electron patterns, leading to comparable reactivity.
Because the table’s layout accommodates each newly discovered element in a specific slot, the total number of slots that have been filled directly corresponds to the count of confirmed elements—currently 118.
Scientific Explanation of the 118 Elements
Electron Shells and Subshells
At the core of the periodic table’s logic is the quantum mechanical model of the atom. Electrons occupy energy levels called shells, which are further divided into subshells labeled s, p, d, and f. The order in which these subshells fill determines an element’s position:
- 1s → Hydrogen (1) and Helium (2)
- 2s, 2p → Lithium (3) through Neon (10)
- 3s, 3p, 3d → Sodium (11) through Krypton (36)
- 4s, 4p, 4d, 4f → Rubidium (37) through Radon (86)
- 5s, 5p, 5d, 5f → Francium (87) through Oganesson (118)
When a new subshell begins to fill, a new period starts, and the table expands accordingly. The completion of the 5f subshell marked the start of the actinide series, while the 6p and 7p subshells introduced the transactinide elements, culminating in element 118.
Stability and Half‑Life
Heavier elements tend to be radioactive, with half‑lives ranging from fractions of a second to millions of years. The stability of an element is influenced by the ratio of protons to neutrons and the presence of “magic numbers” that confer extra nuclear stability. For instance, Copernicium (112) and Flerovium (114) exhibit unusually long half‑lives for their mass numbers, hinting at an “island of stability” that researchers continue to explore.
Classification of the 118 Elements
| Category | Elements Included | Key Characteristics |
|---|---|---|
| Metals | ~80% of the table (e.g., Fe, Cu, Au) | Conductive, malleable, typically solid at room temperature |
| Non‑metals | ~10% (e.g., O, N, C) | Poor conductors, varied physical states |
| Metalloids | ~7 (e.g., Si, Ge) | Intermediate electrical properties, crucial for semiconductors |
| Noble Gases | 7 (He, Ne, Ar, Kr, Xe, Rn, Og) | Chemically inert, full valence shells |
| Halogens | 7 (F, Cl, Br, I, At, Ts, Ts) | Highly reactive non‑metals, form salts |
| Alkali Metals | 6 (Li, Na, K, Rb, Cs, Fr) | Soft, highly reactive, +1 oxidation state |
| Alkaline Earth Metals | 6 (Be, Mg, Ca, Sr, Ba, Ra) | Reactive, +2 oxidation state |
| Transition Metals | 38 (Sc to Cu, etc.) | Variable oxidation states, colorful compounds |
| Inner Transition Metals | Lanthanides (57‑71) and Actinides (89‑103) | f‑block elements, often radioactive |
This classification helps educators and learners navigate the table’s vast array of elements, making it easier to predict chemical behavior.
Historical Perspective: From a Blank Space to 118
When Dmitri Mendeleev first published his periodic table in 1869, only 63 elements were known. He left gaps for undiscovered elements, confidently predicting their properties. Over the next century, each gap was filled, expanding the count and refining the table’s structure. The discovery of synthetic elements in the mid‑20th century—starting with technetium (43) and plutonium (94)—proved that elements could be created artificially, pushing the boundaries beyond naturally occurring substances.
The modern era of element hunting involves particle accelerators and high‑energy nuclear reactions. Teams in Russia, the United States, and Japan have collaborated to synthesize superheavy elements by bombarding heavy target nuclei with lighter projectiles, often producing just a handful of atoms before they decay. The quest for element 119 and beyond continues, with researchers aiming to explore the hypothesized island of stability that might yield longer‑lived super
Continuing from the point aboutthe quest for element 119 and the island of stability:
Synthesis and Challenges: The creation of these superheavy elements is an extraordinary feat of modern science. Researchers employ sophisticated particle accelerators to collide heavy target nuclei, such as those of berkelium or californium, with even heavier projectiles like calcium-48 ions. The goal is a rare, head-on collision where the nuclei fuse, forming a new, heavier nucleus. However, the probability of such fusion is vanishingly small, and the resulting compound nucleus is often highly unstable, decaying almost instantaneously via fission or alpha decay. Detecting and identifying these fleeting atoms requires incredibly sensitive detectors and sophisticated data analysis, often requiring months or years to confirm a single new element.
The Island of Stability: A Beacon of Hope: The island of stability remains the holy grail of superheavy element research. Theoretical models predict that certain combinations of protons and neutrons (magic numbers) could confer extraordinary stability, potentially leading to isotopes with half-lives long enough to allow for chemical study. While the exact location and existence of this island are still debated, the synthesis of elements like Copernicium (112) and Flerovium (114), which showed unexpectedly longer half-lives than many of their neighbors, provided tantalizing hints. Discovering and characterizing elements within this island would revolutionize our understanding of nuclear structure and the limits of the periodic table.
Beyond 118: The Future of the Table: The discovery of element 118, Oganesson, completed the seventh row, but the journey is far from over. The pursuit of elements 119 and 120, and potentially beyond, continues with renewed vigor. New targets and projectiles are being explored, and novel experimental techniques are being developed. The synthesis of these elements pushes the boundaries of human technological capability, requiring advancements in accelerator design, target materials, and detection systems. Each new element synthesized not only adds a name to the table but also provides crucial data points to test and refine our fundamental theories of atomic nuclei and the forces that bind them.
Conclusion:
The periodic table, conceived by Mendeleev as a framework for known elements, has evolved into a dynamic map charting the frontiers of human knowledge. From its humble beginnings with 63 elements and deliberate gaps, it has grown to encompass 118 confirmed elements, including a growing family of synthetic superheavy elements forged in the heart of particle accelerators. This journey, driven by relentless curiosity and cutting-edge technology, has transformed the table from a static catalog into a living document of discovery. The ongoing quest to synthesize elements beyond 118, particularly those tantalizingly predicted to reside on the island of stability, embodies the spirit of exploration that defines science. Each new element synthesized not only expands the table's physical boundaries but also deepens our understanding of the fundamental forces governing matter itself. The periodic table, thus, stands not merely as a classification system, but as a testament to humanity's enduring drive to uncover the hidden structures of the universe, one element at a time.
Latest Posts
Latest Posts
-
Compare Contrast Animal And Plant Cells
Mar 14, 2026
-
Lowest Common Multiple Of 3 And 6
Mar 14, 2026
-
What Is The Difference Between Heterozygous And Homozygous Individuals
Mar 14, 2026
-
What Is The Difference Between Asexual And Sexual Reproduction
Mar 14, 2026
-
How Many Miles Is 42 Km
Mar 14, 2026
Related Post
Thank you for visiting our website which covers about The Periodic Table Is Made Of _____ Known Elements. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.