Molecular Mass Of Ca No3 2
sampleletters
Mar 16, 2026 · 6 min read
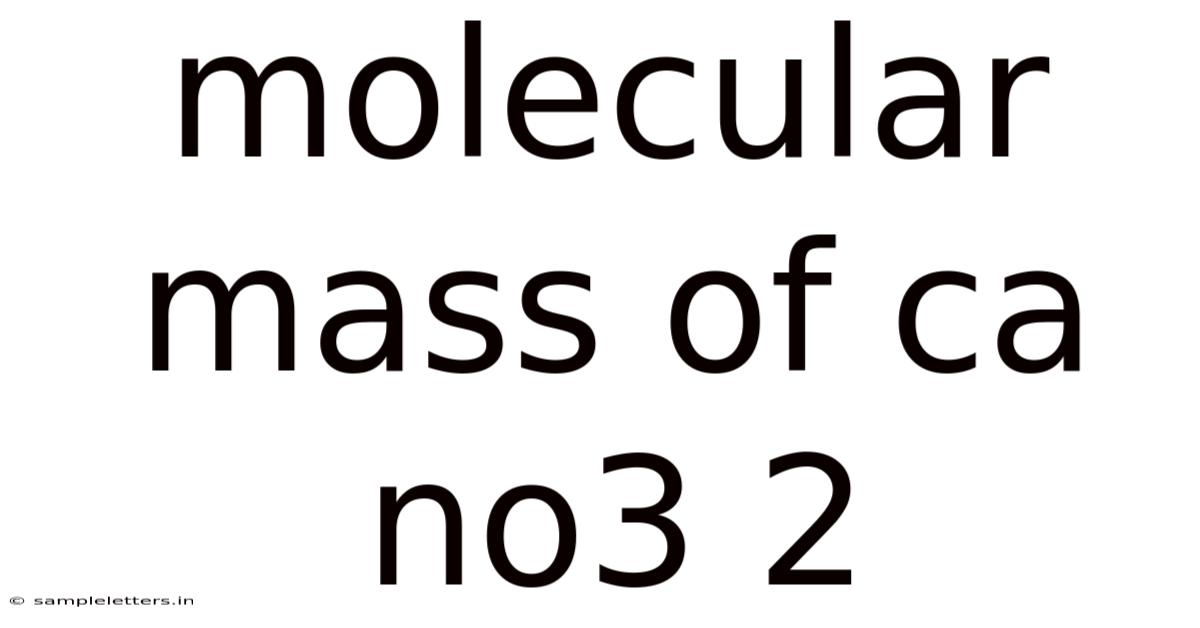
Table of Contents
The molecular mass of a compound is a fundamental concept in chemistry, serving as a critical bridge between the microscopic world of atoms and the measurable quantities used in laboratories and industries worldwide. For calcium nitrate, with the chemical formula Ca(NO₃)₂, calculating its molecular mass is a precise exercise that reveals the weight of one mole of its constituent particles. This value, expressed in grams per mole (g/mol), is not merely an abstract number; it is the key to stoichiometric calculations, solution preparation, and understanding the composition of this widely used inorganic salt. Accurately determining the molecular mass of Ca(NO₃)₂ requires a clear understanding of atomic masses, formula interpretation, and careful arithmetic.
Understanding the Building Blocks: Atomic Mass and Formula Units
Before performing any calculation, we must define our terms. The atomic mass of an element, found on the periodic table, represents the weighted average mass of all naturally occurring isotopes of that atom, relative to 1/12th the mass of a carbon-12 atom. It is listed in atomic mass units (amu) but is numerically identical to the mass in grams of one mole of that element. For our calculation, we will use the standard atomic masses:
- Calcium (Ca): 40.08 amu (or g/mol)
- Nitrogen (N): 14.01 amu (or g/mol)
- Oxygen (O): 16.00 amu (or g/mol)
The formula Ca(NO₃)₂ requires careful parsing. It indicates one atom of calcium (Ca) and two polyatomic nitrate ions (NO₃⁻). The subscript '2' outside the parentheses applies to everything inside it. Therefore, the total number of atoms in one formula unit is:
- 1 Calcium (Ca) atom
- 2 Nitrogen (N) atoms (because 2 × NO₃ contains 2 N)
- 6 Oxygen (O) atoms (because 2 × NO₃ contains 2 × 3 = 6 O)
This interpretation is the most common point of error, making it essential to expand the formula mentally or on paper to CaN₂O₆ before summing the masses.
Step-by-Step Calculation of the Molecular Mass
With the atom counts confirmed, the calculation becomes a straightforward summation of the contributions from each element.
-
Mass contribution from Calcium (Ca):
- Number of atoms: 1
- Atomic mass: 40.08 g/mol
- Total: 1 × 40.08 = 40.08 g/mol
-
Mass contribution from Nitrogen (N):
- Number of atoms: 2
- Atomic mass: 14.01 g/mol
- Total: 2 × 14.01 = 28.02 g/mol
-
Mass contribution from Oxygen (O):
- Number of atoms: 6
- Atomic mass: 16.00 g/mol
- Total: 6 × 16.00 = 96.00 g/mol
-
Summing the contributions:
- Molecular Mass = (Mass from Ca) + (Mass from N) + (Mass from O)
- Molecular Mass = 40.08 g/mol + 28.02 g/mol + 96.00 g/mol
- Molecular Mass = 164.10 g/mol
Therefore, the molecular mass of calcium nitrate, Ca(NO₃)₂, is 164.10 grams per mole. This means that one mole (6.022 × 10²³ formula units) of pure Ca(NO₃)₂ has a mass of exactly 164.10 grams. The precision of this value depends on the atomic masses used; slight variations may occur if using older or less precise periodic tables, but 164.10 g/mol is the standard accepted value.
Common Pitfalls and How to Avoid Them
Several frequent mistakes can lead to an incorrect molecular mass:
- Misreading the Subscript: The most typical error is forgetting to multiply the oxygen count by the outer subscript '2'. Remember, the formula is not CaNO₃; it is Ca(NO₃)₂. Always expand the formula first: CaN₂O₆.
- Using Atomic Numbers Instead of Atomic Masses: Atomic number (proton count) is irrelevant for mass calculations. Always refer to the atomic mass or weight on the periodic table.
- Incorrect Rounding: While atomic masses are often given to two decimal places, intermediate sums should retain these decimals to avoid rounding errors in the final total. Only round the final answer to two decimal places, consistent with the least precise atomic mass used (oxygen at 16.00 has two decimal places).
- Confusing Molecular Mass with Molar Mass: For a covalent compound like Ca(NO₃)₂ (which is actually ionic but we treat its formula unit the same way), the terms molecular mass and formula mass are often used interchangeably. Molar mass is the same numerical value but explicitly carries the unit g/mol, emphasizing its use for macroscopic quantities.
Why This Number Matters: Real-World Applications
Knowing the exact molecular mass of Ca(NO₃)₂ is not an academic exercise. It has direct, practical implications:
- Agricultural Chemistry: Calcium nitrate is a vital fertilizer providing both calcium and nitrogen, two essential plant nutrients. Farmers and agronomists use its 164.10 g/mol molar mass to precisely calculate application rates. To deliver a specific amount of nitrogen (N), they must convert the desired nitrogen mass back to the required mass of Ca(NO₃)₂ using the mass ratios derived from the molar mass.
- Laboratory Preparation: A chemist needing to make a 1-liter, 0.5 M solution of calcium nitrate must weigh out 0.5 mol × 164.10 g/mol = 82.05 grams of the salt. An error in the molecular mass calculation would lead to an incorrect solution concentration, potentially ruining sensitive experiments or analyses.
- Industrial Manufacturing: In the production of concrete admixtures,
calcium nitrate is used to accelerate setting time and improve workability. Precise stoichiometric calculations, relying on the accurate molar mass, are crucial for achieving the desired performance characteristics of the concrete. Deviation from the correct mass can compromise the strength and durability of the final product.
- Pharmaceutical Applications: Calcium nitrate finds use in medicine, particularly as a vasodilator to treat conditions like angina. The precise dosage of calcium nitrate administered to a patient is meticulously calculated based on its molar mass to ensure therapeutic efficacy and minimize side effects.
Conclusion
The molecular mass of Ca(NO₃)₂ is a fundamental concept in chemistry, providing a crucial link between the microscopic world of atoms and molecules and the macroscopic world of measurable quantities. Understanding how to calculate it accurately, and being aware of potential pitfalls, is essential for a wide range of applications, from ensuring optimal crop yields to developing life-saving medications. While seemingly a simple calculation, the accurate determination of molecular mass underscores the importance of precision and careful attention to detail in chemical calculations – a skill vital for success in scientific endeavors and practical industries alike. The reliable molar mass of Ca(NO₃)₂ empowers us to translate theoretical knowledge into tangible, real-world solutions, highlighting its enduring significance in both academic and applied chemistry.
Conclusion
The molecular mass of Ca(NO₃)₂ is a fundamental concept in chemistry, providing a crucial link between the microscopic world of atoms and molecules and the macroscopic world of measurable quantities. Understanding how to calculate it accurately, and being aware of potential pitfalls, is essential for a wide range of applications, from ensuring optimal crop yields to developing life-saving medications. While seemingly a simple calculation, the accurate determination of molecular mass underscores the importance of precision and careful attention to detail in chemical calculations – a skill vital for success in scientific endeavors and practical industries alike. The reliable molar mass of Ca(NO₃)₂ empowers us to translate theoretical knowledge into tangible, real-world solutions, highlighting its enduring significance in both academic and applied chemistry. Furthermore, advancements in analytical techniques, such as mass spectrometry, continually refine our ability to determine molecular masses with increasing accuracy, bolstering the reliability of these calculations across all disciplines. Ultimately, appreciating the significance of this seemingly basic value reveals a cornerstone of scientific understanding and a key to countless practical advancements shaping our world.
Latest Posts
Latest Posts
-
What Are The Factors Of 3
Mar 16, 2026
-
What Is 75 Inches In Feet
Mar 16, 2026
-
How Many Inches Are 15 Cm
Mar 16, 2026
-
Binary Tree Vs Binary Search Tree
Mar 16, 2026
-
How Long Does It Take A Giraffe To Puke
Mar 16, 2026
Related Post
Thank you for visiting our website which covers about Molecular Mass Of Ca No3 2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.