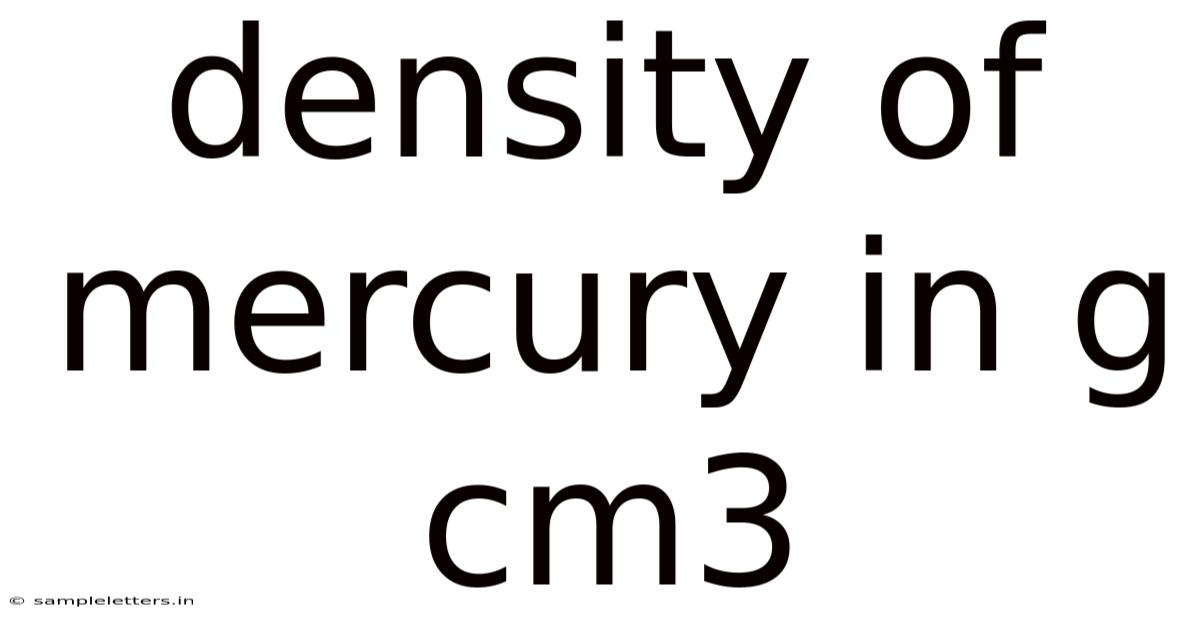Mercury, the only metal that remains liquid at room temperature, is renowned for its high density, a property that makes it indispensable in various scientific and industrial applications. The density of mercury in g cm3 is a fundamental characteristic that influences its behavior in thermometers, barometers, and countless laboratory techniques. Understanding this value, how it is measured, and why it matters provides a gateway to deeper insights into fluid mechanics, material science, and environmental health. This article explores the numerical value of mercury’s density, the methods used to determine it, the underlying scientific principles, and answers to common questions that arise when studying this unique substance It's one of those things that adds up..
Introduction
The density of mercury in g cm3 is approximately 13.And 534 g cm⁻³ at 25 °C (77 °F). In practice, this figure represents the mass of mercury contained in a cubic centimeter of the liquid metal under standard conditions. Because of that, because mercury is denser than most common materials, it sinks in water and many other liquids, a property that is exploited in devices such as mercury-in-glass thermometers and in the separation of precious metals from ore. That's why the precise measurement of mercury’s density is also crucial for calibrating instruments, ensuring accurate volume‑to‑mass conversions, and assessing environmental contamination. The following sections outline how this density is determined, the science behind it, and address frequently asked questions That's the whole idea..
No fluff here — just what actually works.
How the Density Is Measured
Laboratory Techniques
-
Pyccnometer Method – A pyccnometer is a precisely calibrated glass vessel that can hold a known volume of liquid. By weighing the empty pycnometer, then the pycnometer filled with mercury, and finally the pycnometer filled with a reference liquid of known density, the density of mercury can be calculated using the formula:
[ \rho_{\text{Hg}} = \frac{m_{\text{Hg}} - m_{\text{empty}}}{V_{\text{Hg}}} ]
where (m_{\text{Hg}}) is the mass of mercury, (m_{\text{empty}}) is the mass of the empty pycnometer, and (V_{\text{Hg}}) is the known volume of the pycnometer.
-
Hydrostatic Balance – This method employs a balance that measures the buoyant force acting on a submerged object. A small sphere of known volume is weighed in air and then immersed in mercury. The difference in weight provides the mass of mercury displaced, allowing the density to be derived.
-
Digital Density Meters – Modern instruments use oscillating tubes or ultrasonic sensors to determine density directly. The device measures the frequency change caused by the added mass of mercury, converting this signal into a density reading with high accuracy.
Practical Considerations
- Temperature Control – Mercury’s density varies with temperature; it decreases as temperature rises. Which means, measurements are typically reported at 25 °C, and corrections are applied for other temperatures using empirical expansion coefficients.
- Purity of Sample – Trace impurities (e.g., other metals or dissolved gases) can slightly alter the measured density, so high‑purity mercury is essential for precise results.
- Safety Precautions – Because mercury is toxic, all measurements must be performed in well‑ventilated areas with appropriate protective equipment to prevent inhalation or skin contact.
Scientific Explanation of Mercury’s High Density
The density of mercury in g cm3 stems from its atomic structure and bonding characteristics. Mercury atoms are relatively heavy (atomic mass ≈ 200.59 u) and are arranged in a close‑packed, metallic lattice. On top of that, unlike most metals, mercury’s atoms are held together by weak van der Waals forces rather than strong metallic bonds, resulting in a liquid state at ambient conditions. That said, despite the weak bonding, the large atomic mass and relatively small atomic radius allow mercury atoms to pack closely together, maximizing mass per unit volume. This combination of high atomic weight and efficient packing yields a density that surpasses that of most other liquids, including water (1 g cm⁻³) and many organic solvents It's one of those things that adds up..
The underlying physics can be visualized as a densely packed crowd of heavy individuals occupying minimal space, which translates into a high mass per unit volume.
Additionally, the relativistic effects on mercury’s electrons influence its metallic bonding, further contributing to its compact arrangement and high density That's the part that actually makes a difference..
Frequently Asked Questions (FAQ)
Q1: Why does mercury have a higher density than water?
A: Mercury’s atoms are much heavier than water molecules, and they pack more efficiently in the liquid state, resulting in a mass‑to‑volume ratio that is roughly 13.5 times greater than that of water.
Q2: Does the density of mercury in g cm3 change with altitude?
A: The density change due to altitude is negligible because pressure variations affect mercury only minimally; however, temperature fluctuations have a more noticeable impact.
Q3: How does the density of mercury in g cm3 compare to other metals?
A: Mercury is denser than most common metals such as aluminum (2.70 g cm⁻³), iron (7.87 g cm⁻³), and copper (8.96 g
cm⁻³), but less dense than tungsten (19.3 g cm⁻³) and osmium (22.59 g cm⁻³).
Q4: Is the density of mercury in g cm3 affected by pressure?
A: Yes, like all liquids, mercury’s density increases with pressure. Still, the effect is small compared to solids and is typically negligible in everyday applications.
Q5: Why is mercury used in thermometers despite its high density?
A: Mercury’s high density allows for a compact and sensitive instrument, but its unique property of expanding with temperature change makes it ideal for measuring temperature. Additionally, its silvery appearance and ease of visual tracking make it practical for thermometric use Practical, not theoretical..
Applications of Mercury’s Density
Mercury’s density and physical properties have led to its use in various applications, though many of these are now being phased out due to environmental and health concerns. Some of the key applications include:
- Barometers and Barometric Pressure Measurement – Mercury barometers use the liquid’s density to measure atmospheric pressure accurately.
- Thermometers – Historically, mercury thermometers were widely used for their precision and reliability, especially in medical and laboratory settings.
- Buoyancy and Density Measurement – Mercury’s density is used in hydrometers and pycnometers to measure the densities of other liquids.
- Industrial Applications – Mercury is used in certain types of electrical switches, generators, and as a coolant in specialized equipment due to its high thermal conductivity and density.
Environmental and Health Considerations
Despite its utility, mercury’s toxicity has led to strict regulations on its use and disposal. Plus, the Minamata Convention on Mercury, an international treaty, aims to protect human health and the environment from mercury pollution by reducing its use and exposure. This leads to many countries have banned or restricted the use of mercury in thermometers, switches, and other products, pushing for alternatives such as digital thermometers and other non-toxic materials Worth knowing..
Conclusion
The density of mercury in g cm3 is a remarkable physical property that has both fascinated scientists and served practical applications throughout history. With a density of approximately 13.6 g cm⁻³ at 20 °C, mercury’s unique characteristics make it a valuable substance, albeit one that requires careful handling due to its toxicity. As science and technology evolve, the focus on safer and more sustainable alternatives continues to grow, reflecting a broader shift towards environmental stewardship and public health protection. Nonetheless, the study of mercury’s density remains a fundamental aspect of physics and chemistry, offering insights into the behavior of matter and the forces that govern our universe.
Not the most exciting part, but easily the most useful.
Conclusion
At the end of the day, while mercury’s density and other properties have made it a significant player in various scientific and industrial applications, the growing awareness of its environmental and health risks has led to a global effort to phase it out. Worth adding: this shift is not just about finding safer alternatives but also about fostering a deeper understanding of how elements interact with the world around us. As we move forward, the legacy of mercury serves as a reminder of the delicate balance between technological advancement and environmental responsibility. The future of chemistry and physics lies in embracing innovations that reduce harm, ensuring that our quest for knowledge does not come at the cost of our planet’s health Turns out it matters..